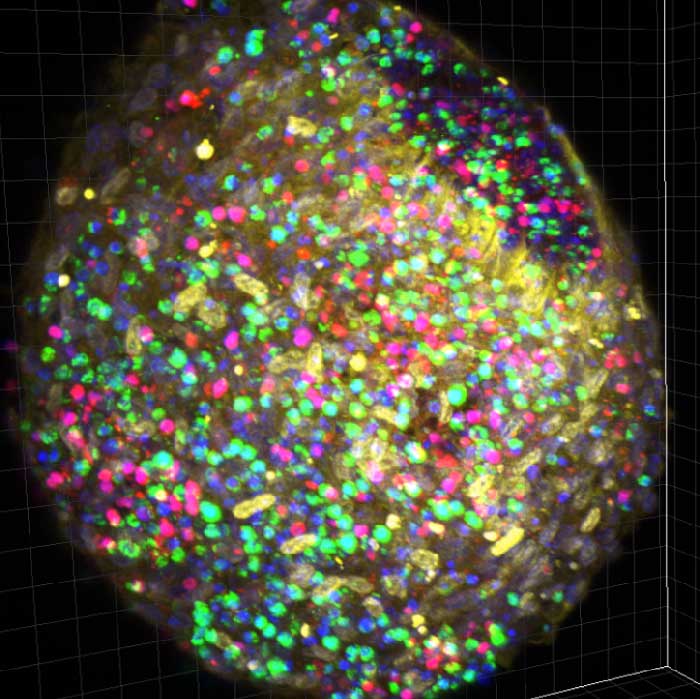
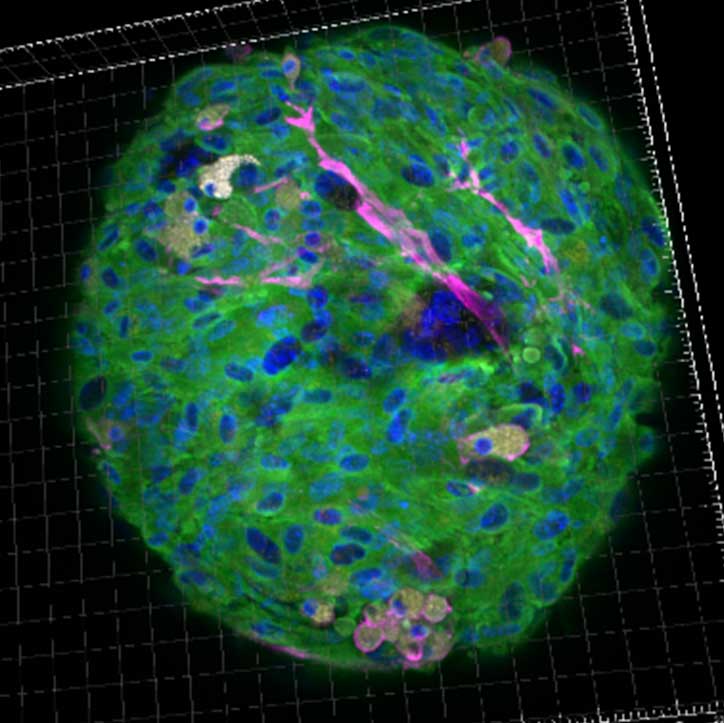
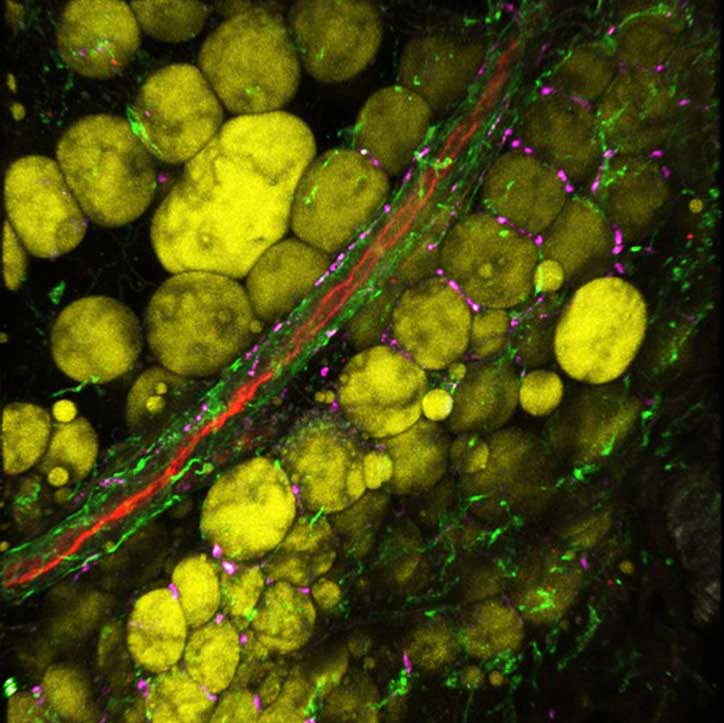
FLAMES
Agnès Coste / Nicolas Espagnolle
FLAMES : INFLAMMAGING CONTROL BY DYNAMIC MSC / MYELOID CELL CROSSTALK
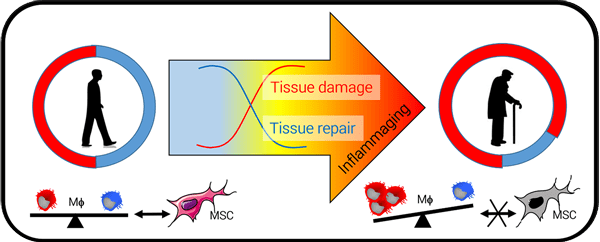
The objective of the FLAMES team is to integrate stromal dynamics, inflammation and metabolism in the understanding of the loss of biological functions during aging to maintain and/or restore functional capacities. Our project aims to demonstrate that age-related chronic inflammation results from a defect of resolution of inflammation in tissues due to altered dialogue between macrophages and mesenchymal stromal cells (MSC).
This project is based on the use of innovative animal models, in vitro 4D strategies to reproduce tissue heterogeneity, as well as in silico approaches to study the inflammatory response and their effects on tissue repair.
The goal of our research project is to contribute to the identification of potential therapeutic targets to restore the dialogue between MSCs and macrophages to promote the development of a pro-resolutive response.
research axis
AXIS 1
Mapping of Macrophages and MSC heterogeneity during physiological and pathophysiological aging
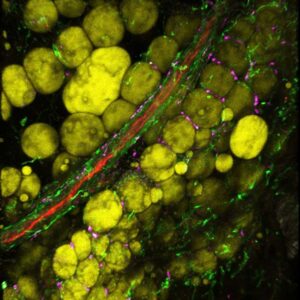 Our group are looking at the modification of the phenotype and of the modification of the functional capacities of macrophages and MSC during aging in human and in mice. This will be done by using confocal microscopy, flow cytometry and single cell transcriptomic approaches, as well as functional assays. In addition, as the macrophages heterogeneity relies also on their origin, the modification of the macrophage origin during aging are studying using lineage tracing and animal models of hematopoietic reconstruction.
Our group are looking at the modification of the phenotype and of the modification of the functional capacities of macrophages and MSC during aging in human and in mice. This will be done by using confocal microscopy, flow cytometry and single cell transcriptomic approaches, as well as functional assays. In addition, as the macrophages heterogeneity relies also on their origin, the modification of the macrophage origin during aging are studying using lineage tracing and animal models of hematopoietic reconstruction.
AXIS 2
Identify cellular and molecular targets that control the MSC/ Macrophages crosstalk during aging
Our group is characterizing the receptors and soluble factors involved in the interaction between MSC and macrophages and how this MSC-macrophage communication is modified during aging. We currently focus on CD54 as a key molecule in the cross-talk between Macrophages and MSCs (Esapgnolle et al) and on ecosanoids that play a central role in resolution of inflammation (ref).
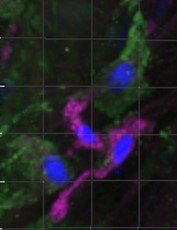
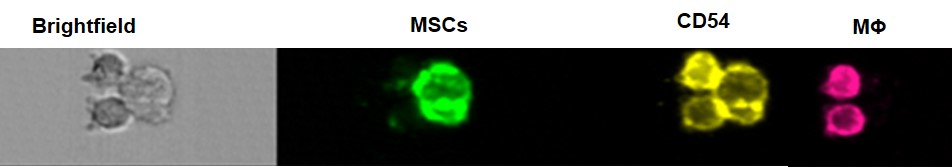
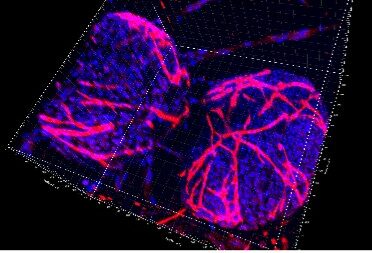
The targets involved in MSC-macrophage interaction are studying using murine and human experimental models of vascularized 3D culture systems that combine different cellular actors
- endothelial cells
- immune cells
- stroma
AXIS 3
Strategies to restore the pro-resolutive macrophages through the re-education of macrophages or MSC/ Macrophages crosstalk
Our group is also developing different therapeutic approaches to reduce “inflammaging”. Pharmacological, nutritional approaches and also cell therapy approaches will be developed.
This research is carried out in collaboration with university partners and private companies.
team members
Publications
- Luche E, Robert V, Cuminetti V, Pomié C, Sastourné-Arrey Q, Waget A, Arnaud E, Varin A, Labit E, Laharrague P, Burcelin R, Casteilla L, Cousin B. Corrupted adipose tissue endogenous myelopoiesis initiates diet-induced metabolic disease. Elife. (2017) 28;6.
- Espagnolle N, Balguerie A, Arnaud E, Sensebé L, Varin A. CD54-Mediated Interaction with Pro-inflammatory Macrophages Increases the Immunosuppressive Function of Human Mesenchymal Stromal Cells. Stem Cell Reports. (2017), 8(4):961-976.
- Martini H, Iacovoni JS, Maggiorani D, Dutaur M, Marsal DJ, Roncalli J, Itier R, Dambrin C, Pizzinat N, Mialet-Perez J, Cussac D, Parini A, Lefevre L, Douin-Echinard V. Aging induces cardiac mesenchymal stromal cell senescence and promotes endothelial cell fate of the CD90 + subset. Aging Cell. (2019), 18(5):e13015.
- Lefèvre L, Authier H, Stein S, Majorel C, Couderc B, Dardenne C, Eddine MA, Meunier E, Bernad J, Valentin A, Pipy B,Schoonjans K, Coste A. LRH-1 mediates anti-inflammatory and antifungal phenotype of IL-13-activated macrophages through the PPAR ligand synthesis. Nat Commun. (2015), 6:6801.
- Alaeddine M, Prat M, Poinsot V, Gouazé-Andersson V, Authier H, Meunier E, Lefèvre L, Alric C, Dardenne C, Bernad J, Alric L, Segui B, Balard P, Couderc F, Couderc B, Pipy B, Coste A. IL13-Mediated Dectin-1 and Mannose Receptor Overexpression Promotes Macrophage Antitumor Activities through Recognition of Sialylated Tumor Cells. Cancer Immunol Res. (2019),7(2):321-334
- Rahabi M, Jacquemin G, Prat M, Meunier E, AlaEddine M, Bertrand B, Lefèvre L, Benmoussa K, Batigne P, Aubouy A, Auwerx J, Kirzin S, Bonnet D, Danjoux M, Pipy B, Alric L, Authier H, Coste A. Divergent Roles for Macrophage C-type Lectin Receptors, Dectin-1 and Mannose Receptors, in the Intestinal Inflammatory Response. Cell Rep. (2020), 30(13):4386-4398
- Prat M, Le Naour A, Coulson K, Lemée F, Leray H, Jacquemin G, Rahabi MC, Lemaitre L, Authier H, Ferron G, Barret JM, Martinez A, Ayyoub M, Delord JP, Gladieff L, Tabah-Fisch I, Prost JF, Couderc B, Coste A. Circulating CD14high CD16low intermediate blood monocytes as a biomarker of ascites immune status and ovarian cancer progression. J Immunother Cancer. (2020), 8(1):e000472
- Espagnolle N, Hebraud B, Descamps JG, Gadelorge M, Joubert MV, Ferreira LDS, Roussel M, Huynh A, Sensébé L, Casteilla L, Attal M, Avet-Loiseau H, Deschaseaux F, Bourin P, Corre J. Functional Comparison between Healthy and Multiple Myeloma Adipose Stromal Cells. Stem Cells Int. (2020), 4173578.
- Prat M, Salon M, Allain T, Dubreuil O, Noël G, Preisser L, Jean B, Cassard L, Lemée F, Tabah-Fish I, Pipy B, Jeannin P, Prost JF, Barret JM, Coste A. Murlentamab, a Low Fucosylated Anti-Müllerian Hormone Type II Receptor (AMHRII) Antibody, Exhibits Anti-Tumor Activity through Tumor-Associated Macrophage Reprogrammation and T Cell Activation. Cancers. (2021), 13(8):1845.
- Martini H, Lefevre L, Sayir S, Itier R, Maggiorani D, Dutaur M, Marsal DJ, Roncalli J, Pizzinat N, Cussac D, Parini A, Mialet-Perez J, Douin-Echinard V. Selective Cardiomyocyte Oxidative Stress Leads to Bystander Senescence of Cardiac Stromal Cells. Int J Mol Sci. (2021), 22(5):2245.
- Lefèvre L, Iacovoni JS, Martini H, Bellière J, Maggiorani D, Dutaur M, Marsal DJ, Decaunes P, Pizzinat N, Mialet-Perez J, Cussac D, Parini A, Douin-Echinard V. Kidney inflammaging is promoted by CCR2+ macrophages and tissue-derived micro-environmental factors. Cell Mol Life Sci. (2021), 78(7):3485-3501
- Rabiller L, Robert V, Arlat A, Labit E, Ousset M, Salon M, Coste A, Da Costa-Fernandes L, Monsarrat P, Ségui B, André M, Guissard C, Renoud ML, Silva M, Mithieux G, Raymond-Letron I, Pénicaud L, Lorsignol A, Casteilla L, Dromard Berthézène C, Cousin B. Driving regeneration, instead of healing, in adult mammals: the decisive role of resident macrophages through efferocytosis. NPJ Regen Med. (2021), 6(1):41.
- Rahabi M, Salon M, Bruno-Bonnet C, Prat M, Jacquemin G, Benmoussa K, Alaeddine M, Parny M, Bernad J, Bertrand B, Auffret Y, Robert-Jolimaître P, Alric L, Authier H, Coste A. Bioactive fish collagen peptides weaken intestinal inflammation by orienting colonic macrophages phenotype through mannose receptor activation. Eur J Nutr. (2022).
- Lemaitre L, Hamaidia M, Descamps JG, Do Souto Ferreira L, Joubert MV, Gadelorge M, Avet-Loiseau H, Justo A, Reina N, Deschaseaux F, Martinet L, Bourin P, Corre J, Espagnolle N. Toll-like receptor 4 selective inhibition in medullar microenvironment alters multiple myeloma cell growth. Blood Adv. (2022), 6(2):672-678.
- Dardenne C, Salon M, Authier H, Meunier E, AlaEddine M, Bernad J, Bouschbacher M, Lefèvre L, Pipy B, Coste A. Topical Aspirin Administration Improves Cutaneous Wound Healing in Diabetic Mice Through a Phenotypic Switch of Wound Macrophages Toward an Anti-inflammatory and Proresolutive Profile Characterized by LXA4 Release. Diabetes. (2022), 71(10):2181-2196.
- Prat M, Coulson K, Blot C, Jacquemin G, Romano M, Renoud ML, AlaEddine M, Le Naour A, Authier H, Rahabi MC, Benmoussa K, Salon M, Parny M, Delord JP, Ferron G, Lefèvre L, Couderc B, Coste A. PPARγ activation modulates the balance of peritoneal macrophage populations to suppress ovarian tumor growth and tumor-induced immunosuppression. J Immunother Cancer. (2023), 11(8): e007031.
- Arlat A, Renoud ML, Nakhle J, Thomas M, Fontaine J, Arnaud E, Dray C, Authier H, Monsarrat P, Coste A, Casteilla L, Ousset M, Cousin B. Generation of functionally active resident macrophages from adipose tissue by 3D cultures. Front Immunol. (2024) 15:1356397.
- Blot C, Lavernhe M, Lugo-Villarino G, Coulson K, Salon M, Tertrais M, Planès R, Santoni K, Authier H, Jacquemin G, Rahabi M, Parny M, Letron IR, Meunier E, Lefèvre L, Coste A. Leishmania infantum exploits the anti-ferroptosis effects of Nrf2 to escape cell death in macrophages. Cell Rep. (2024) 43(9):114720.
- Gandolfi S, Sanouj A, Chaput B, Coste A, Sallerin B, Varin A. The role of adipose tissue-derived stromal cells, macrophages and bioscaffolds in cutaneous wound repair. Biology Direct (2024) 19:85
- Soussi S, Maione AS, Lefevre L, Pizzinat N, Iacovoni J, Gonzalez-Fuentes I, Cussac D, Iengo L, Santin T, Tundo F, Tondo C, Pompilio G, Parini A, Douin-Echinard V, Sommariva E. Analysis of effector/memory regulatory T cells from arrhythogenic cardiomyopathy patients identified IL-32 as novel player in ACM pathogenesis. Cell Death Dis (2025) 16(1):87
- Brodeau C, Joly C, Chekroun A, Nakhle J, Blase V, Espagnolle N, Dray C, Yart A, Planat V, Tertais M, Fassy J, Guyonnet S, Lu WH, de Souto Barreto P, Teste O, Tremblay-Franco M, Tanner KT, Cohen AA, Carrière A, Casteilla L, Ader I. Identification of functional cellular markers related to human health, frailty and chronological age. Aging Cell (2025) 24(9): e70153
- Patent N°2854078 Utilisation d’agonistes de PPARgamma comme agents anti-infectieux.
- Patent N° 2984719 Méthode d’analyse de la progression de la cicatrisation d’une plaie à des niveaux macroscopiques et microscopiques;
- Patent N° 2984722 Dispositif non-invasif destiné à recueillir les exsudats d’une plaie, son utilisation et kit comprenant ledit dispositif
- Patent N° 1262644 Utilisation acide acétyl salicylique pour la prévention et/ou le traitement des plaies du diabétique
- Patent N° FR1750280 et extension à l’international en 2019 ” Effets immunomodulateurs du peptide P17″
- Patent FR1663344 Antagoniste spécifique de TLR4 dans le traitement du myélome multiple
- Patent FR2102708 PCT/2022/057229 Medium and process for the production of bone marrow reconstitution.
- Patent FR2302374 Matrice 3D poreuse à base de complexes de poly électrolytes et ses applications
- Patent EP23305138.2 PCT/EP2024/052113 Compound for use in the prevention and/or treatment of cancer
- Patent EP23306476.5 Processes for producing a reconstructed skin model, and applications thereof
- Patent FR2209990; WO/2024/068960 Process for producing a three-dimensional model of human multiple myeloma

































