METABOLINK
Cédric Dray / Armelle Yart
Metabolic interplays and age-related loss of function
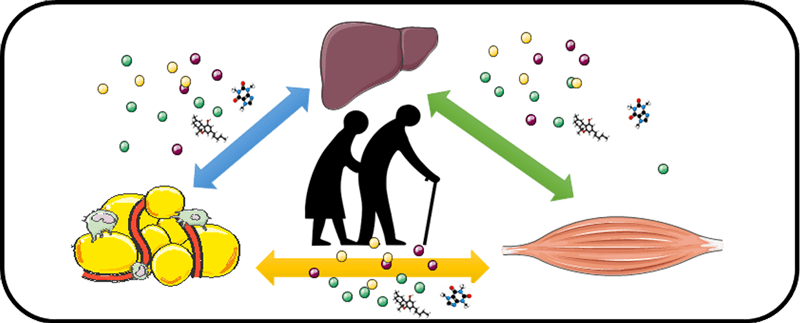
During lifespan, environmental fluctuations consistently forced organism and cells to develop metabolic flexibility in order to maintain their energy homeostasis.
In this context, many aspects of cellular/tissue metabolism changes are driven by redox metabolism as well as inter-cellular and inter-organs metabolic interactions that are fundamental to maintain tissue and whole body energetic homeostasis. Altogether, these parameters are in constant adaptation and represent the metabolic resilience of cell (cell capacity to face metabolic alterations).
We hypothesized that metabolic resilience directs the ability of the aging organism to maintain the main metabolic functions (storage, utilization and release of energy) necessary to perform towards healthy-aging or dependence when disrupted.
Our aim is to investigate the role and the regulation of local/systemic metabolic dialogue within a tissue (mesenchyma/parenchyma) and between metabolic tissues (adipose tissues/muscle/liver) by focusing on intracellular changes in metabolic pathways and redox state triggering major modifications of secretion and cell production. This will allow identifying new mechanisms that contribute, when defective, to the onset or aggravation of age-related diseases (sarcopenia, metabolic diseases) that could represent therapeutic targets of interest.
research areas
AXIS 1
Metabokines and age-related loss of function
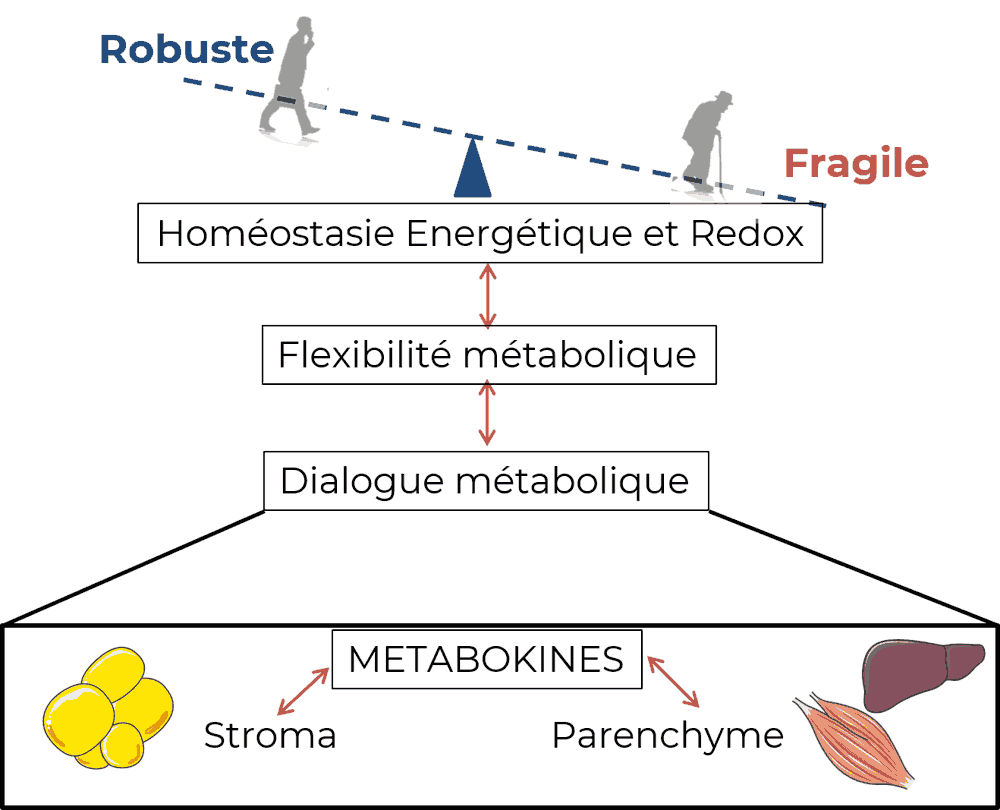
 Inter-cellular and inter-organs metabolic interactions are fundamental to maintaining tissue and whole body energetic homeostasis. The effectors mediating these metabolic interplays include both hormones/cytokines and metabolites (gathered under the term “metabokines”), the latter being able to act either as nutrients or signaling molecules.
Inter-cellular and inter-organs metabolic interactions are fundamental to maintaining tissue and whole body energetic homeostasis. The effectors mediating these metabolic interplays include both hormones/cytokines and metabolites (gathered under the term “metabokines”), the latter being able to act either as nutrients or signaling molecules.
During aging, these changes of “metabokines” profile could interfere with the dialogue between metabolic tissues such as adipose tissues, muscle and liver and consequently support the decline of metabolic flexibility during aging.
Therefore, the impact of aging on the overall distribution of “metabokines” fluxes remains to be fully described. To that aim, we are developing new models of accelerated of early aging (Nothobranchuis Furzeri, genetic diseases, nutritional input) combined to state-of-the-art approaches (fluxomics, organ-on-chip) able to identify and target new pathways and therapeutic candidates.
AXE 2
L'état redox, un nouveau déclencheur du vieillissement
 Using up to date experimental animal models and human resources, we are establishing the link between age-related loss of metabolic functions and redox state and are studying the causes and consequences of metabolic flexibility alterations during aging in metabolic tissues.
Using up to date experimental animal models and human resources, we are establishing the link between age-related loss of metabolic functions and redox state and are studying the causes and consequences of metabolic flexibility alterations during aging in metabolic tissues.
Many aspects of cellular/tissue metabolism are driven by redox metabolism, which corresponds to a complex network of electron transfer. The maintenance of redox homeostasis is of paramount importance for normal cell and tissue functions and increasing evidences demonstrate that the redox state is the main conductor regulating the metabolic reorganization during stress, which is enabled by functional metabolic flexibility.
In addition to support energy homeostasis, redox state drives many cellular functions including cell proliferation, differentiation, senescence as well as autocrine/paracrine/endocrine activity. While redox stress is well known to be associated with aging, Metabolink aims to understand and to target the network and the dynamics of the metabolic and redox reorganizations associated with aging and age-associated pathologies.
team members
Apelin, sarcopenia, ageing, adipokines, myokines, metabolism

Sarcopenia, aging, inflammation, senescence, Noonan syndrom

Metabolism, adipocytes, metabolomic, fluxomic, isotopic tracing in vitro/in vivo, cell culture (hypoxia), mitochondrial activity

Mass spectrophometry Fluxomic, Metabolomic

Spiro Khoury
In vivo functional exploration, metabolic phenotyping, surgery

Anesthesia, reanimation, pain

metabolism, metabolic network, metabolomic, fluxomic, metabolic modelisation

Liver, adipose tissue, macrophages, metabolism, inflammation, ageing

Apelin/APJ, obesity, adipokines, diabetes, ageing

Publications
Morin E, Doumard E, Hartnell LM, Salegi Ansa B, Leduc-Gaudet JP, Quillien A, Nakhle J, Ethuin S, Goudounèche D, Payré B, Soldan V, Balor S, Mattout A, Rouquette J, Dubois L, Sengenès C, Planat V, Casteilla L, Yart A, Dray C, Aligon J, Ferrucci L, Duchesne É, Hussain SNA, Gouspillou G, Formentini L, Mourier A, Baris OR, Valet P, Parpex H, Monsarrat P, Vigneau M, Pradère JP.EMito-Metrix enables automated evaluation of mitochondrial morphology across species.Nat Metab. 2025 Oct 15. doi: 10.1038/s42255-025-01400-z.
Brodeau C, Joly C, Chekroun A, Nakhle J, Blase V, Espagnolle N, Dray C , Yart A, Planat V , Tertrais M, Fassy J, Guyonnet S, Lu WH, de Souto Barreto P, Teste O, Tremblay-Franco M, Tanner K, Cohen AA, Carriere A, Casteilla L, Ader I. Identification of Functional Cellular Markers Related to Human Health, Frailty and Chronological Age. Aging Cell. 2025 Sep;24(9):e70153. doi: 10.1111/acel.70153.
Montané R, Jeanson Y, Lagarde D, Khoury S, Porcher-Bibes L, Nakhle J, Sallese M, Parny M, Raymond-Letron I, Huard E, de Souza RA, Galinier A, Pellerin L, Sore AB, Pradère JP, Moro C, Casteilla L, Yart A, Dray C, Portais JC, Ader I, Carriere A. A brown adipose tissue activity impacts systemic lactate clearance in male mice. J Physiol. 2025 Sep 22. doi: 10.1113/JP288871.
Tremblay-Franco M, Canlet C, Carriere A, Nakhle J, Galinier A, Portais JC, Dray C, Wan-Hsuan P, Bertrand Michel J, Guyonnet S, Rolland Y, Delrieu J, de Souto Barreto P, Vellas B, Penicaud L, Casteilla L, Ader I. Integrative multi metabolomics to early predict cognitive decline among amyloid positive community-dwelling older adults. J Gerontol A Biol Sci Med Sci. 2024 79(5):glae077. doi: 10.1093/gerona/glae077
Farge T, Nakhle J, Lagarde D, Cognet G, Polley N, Castellano R, Nicolau M-L, Bosc C, Sabatier M, Sahal A, Saland E, Jeanson Y, Guiraud N, Boet E, Bergoglio C, Gotanègre M, Mouchel P-L, Stuani L, Larrue C, Moro C, Dray C, Colette Y, Raymond-Letron I, Ader I, Récher C, Sarry J-E, 3 Cabon F, Vergez F, and Carrière A. CD36 drives metastasis and relapse in acute myeloid leukemia. 2023. Cancer Res;83:2824 – 38
Pey V, Doumard E, Komorowski M, Rouget A, Delmas C, Vardon-Bounes F, Poette M, Ratineau V, Dray C, Ader I, Minville V. A locally optimised machine learning approach to early prognostication of long-term neurological outcomes after out-of-hospital cardiac arrest. Digit Health. 2024 Apr 15;10:20552076241234746. doi: 10.1177/20552076241234746
Kemoun P., Ader I., Planat-Benard V., Dray C., Fazilleau N., Monsarrat P., Cousin B., Paupert J., Ousset M., Lorsignol A., Raymond-Letron I., Vellas B., Valet P., Kirkwood T., Beard J., Pénicaud L., Casteilla L. A gerophysiology perspective on healthy ageing. Ageing Res Rev. 2022, 73:101537. Review
Personnaz J., Dortignac A., Piccolo E., Iacovini J.S., Mariette J., Pollizi A., Batut A., Deleruyelle S., Paccoud R., Moreau E., Martins F., Clouaire T., Rayah F., Montagner A., Wahli W., Schwabe R.F., Yart A., Castan-Laurell I., Postic C., Moro C., Legube G., Guillou H., Valet P., Dray C., Pradère J.P. Nuclear HMGB1 regulates liver lipogenesis through negative regulation of liver X receptor. Sci Adv. 2022,8(12):eabg9055
Ader I., Pénicaud L., Andrieu S., Beard J.R., Davezac N., Dray C., Fazilleau N., Gourdy P., Guyonnet S., Liblau R., Parini A., Payoux P., Rampon C., Raymond-Letron I., Rolland Y., De Souto Barreto P., Valet P., Vergnolle N., Sierra F., Vellas B., Casteilla L. Healthy Aging Biomarkers: The INSPIRE’s Contribution. J Frailty Aging. 2021,10(4):313-319
Bullich S., De Souto Barreto P., Dortignac A., He L., Dray C., Valet P., Guiard BP. Apelin controls emotional behavior in age- and metabolic state-dependent manner Psychoneuroendocrinology. 2022,140:105711
Paccoud R., Saint-Laurent C., Piccolo E., Tajan M., Dortignac A., Pereira O., Le Gonidec S., Baba I., Gélineau A., Askia H., Branchereau M., Charpentier J., Personnaz J., Branka S., Auriau J., Deleruyelle S., Canouil M., Beton N., Salles J.P., Tauber M., Weill J., Froguel P., Neel B.G., Araki T., Heymes C., Burcelin R., Castan I., Valet P., Dray C., Gautier E.L., Edouard T., Pradère J.P., Yart A.. SHP2 drives inflammation-triggered insulin resistance by reshaping tissue macrophage populations. Sci Transl Med. 2021;13(591):eabe2587
Lagarde D., Jeanson Y., Portais J.C., Galinier A., Ader I., Casteilla L, Carrière A. Lactate Fluxes and Plasticity of Adipose Tissues: A Redox Perspective. Front Physiol. 2021,12:689747. Review
Lagarde D., Jeanson Y., Barreau C., Moro C., Peyriga L., Cahoreau E., Guissard C., Arnaud E., Galinier A., Bouzier-Sore A.K., Pellerin L., Chouchani E.T,. Pénicaud L., Ader I., Portais J.C., Casteilla L., Carrière A. Lactate fluxes mediated by the monocarboxylate transporter-1 are key determinants of the metabolic activity of beige adipocytes. J Biol Chem. 2021, 296:100137
Stuani L., Sabatier M., Saland E., Cognet G., Poupin N., Bosc C., Castelli F.A., Gales L., Turtoi E., Montersino C., Farge T., Boet E., Broin N., Larrue C., Baran N., Cissé M.Y,. Conti M., Loric S., Kaoma T., Hucteau A., Zavoriti A., Sahal A., Mouchel P.L., Gotanègre M., Cassan C., Fernando L., Wang F., Hosseini M., Chu-Van E., Le Cam L., Carroll M., Selak M.A, Vey N., Castellano R., Fenaille F., Turtoi A., Cazals G., Bories P., Gibon Y., Nicolay B., Ronseaux S., Marszalek J.R., Takahashi K., DiNardo C.D., Konopleva M., Pancaldi V., Collette Y., Bellvert F., Jourdan F., Linares L.K., Récher C., Portais J.C., Sarry J.E. Mitochondrial metabolism supports resistance to IDH mutant inhibitors in acute myeloid leukemia. J.Exp Med. 2021, 218(5):e2020092
Tajan M., Hennequart M., Cheung E.C., Zani F., Hock A.K., Legrave N., Maddocks O.D.K., Ridgway RA, Athineos D., Suárez-Bonnet A., Ludwig R.L., Novellasdemunt L., Angelis N., Li V.S.W., Vlachogiannis G., Valeri N., Mainolfi N., Suri V., Friedman A., Manfredi M., Blyth K., Sansom O.J., Vousden K.H. Serine synthesis pathway inhibition cooperates with dietary serine and glycine limitation for cancer therapy Nat Commun. 2021,12(1):366
Millard U., Schmitt P., Kiefer J.A., Vorholt S., Heux S., Portais J.C. ScalaFlux: A scalable approach to quantify fluxes in metabolic subnetworks. PLoS Comput Biol. 2020,16(4):e1007799.
Vinel C., Lukjanenko I., Batut A., Deleyruelle S., Pradere J.P., Le Gonidec S., Dortignac A., N. Geoffre, Pereira O., S. Karaz, U. Lee, M. Camus, K. Chaoui, E. Mouisel, A. Bigot, V. Mouly, M. Vigneau, A. Pagano, A. Chopard, Pillard F., S. Guyonnet, M. Cesari, O. Schiltz, M. Pahor, J. Feige, B. Vellas, Valet P., DrayC. The exerkine apelin reverses age-associated sarcopenia. Nat. Med., 2018, 24: 1360-137
- I.Ader, Guyonnet S, Vellas B, Casteilla L, Bordeau C. Methods for predicting the health state of a subject from fibroblasts. Ep N°24306153.8
- I.Ader, Casteilla L, Tremblay-Franco M, Canlet C, Vellas B. Methods for predicting cognitive decline in a subject Ep N° 23306392.4
- I. Ader, J. Aligon, S. Cussat-blanc, L. Penicaud, P. Kemoun , D. Bernard, E. Doumard, P. Monsarrat, L. Casteilla. A method for determinig a physiological age of a subject. Ep N°22305353
- L. Mazeyrie, T. Edouard, C. Dray, J.P. Pradère, P.Valet, A.Yart : Use of Shp2 Inhibitors for inhibiting senescence Ep N° 22305331.1
- R.Paccoud, M. Tajan, P.Valet, C.Dray, JP Pradère, A Yart Use of Shp2 Inhibitors for the treatment of insulin resistance Ep N°18306558.0
- P. Gourdy, I. Castan-Laurell, C. Dray, P. Valet. Method and pharmaceutical composition for use in the treatment of diabetes. Ep N°015305422.6
- C. Dray, V. Minville, B. Frances, P. Valet. Method and pharmaceutical composition for the treatment of post-operative cognitive dysfunction. Ep N°16305170.9
- C. Dray, C. Knauf, O. Kunduzova, I. Castan-Laurell, P. Valet. Method dnd pharmaceutical composition for use in the treatment of dysfunction associated with aging. Ep N° 2 785 365

























